Known as the ‘other CO2 problem’, ocean acidification is a global phenomenon that is resulting from increasing levels of carbon dioxide (CO2) being absorbed into the world’s oceans.
Presently, ocean acidification is overshadowed by a wealth of other risks faced by coral reefs. Nonetheless, the process has the potential to be catastrophic in the not too distant future. Indeed, there has already been a monumental change in ocean chemistry and, since the advent of human industrialization, oceans have become 30% more acidic. New evidence is that these changes are now beginning to endanger sea life.
Approximately a third of the CO2 emitted into our atmosphere is absorbed into the ocean. Once in the ocean, it dissolves and combines with seawater to form carbonic acid. This in turn increases ocean acidity.
Increasing acidity has a profound effect on natural biochemical processes. Problematically, acidification lowers the solubility of oceanic waters to important minerals such as calcium carbonate. This means that waters become saturated with these minerals at lower concentrations and therefore can hold less of them.
For shelled marine organisms such as corals or sea snails, this is a real issue. These organisms rely on sequestering calcium carbonate from their surrounding environment in order to produce the exoskeletons which protect them from predation, damage and infection.
The construction of shells in marine animals is dynamic. This means that as organisms synthesise new shell on the inside, older shell is becoming eroded by physical and chemical processes on the outside. In suitable conditions, the growth of new shell outpaces the deterioration of old shell. When ocean acidity increases, this balance is reversed and shells effectively dissolve. Without the protection of an exoskeleton, the organisms may well be driven to extinction.
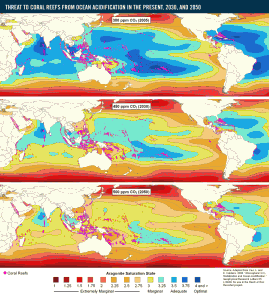
Computer models that predict future trends in ocean acidity have provided us with some alarming insights. These models, based on predictable trends in atmospheric CO2 concentrations, suggest that by the year 2050, aragonite (a form of calcium carbonate) saturation levels globally will be below the optimal level required for coral growth. Even more worryingly, vast swathes of the ocean are predicted to become saturated at such low levels of aragonite that shell formation could not occur at all.
Recent research has identified that some ocean regions are already at this stage and that the shells of living organisms (marine snails in this case) are indeed actively dissolving. These observations, made in polar regions, are considered a result of anthropogenically produced atmospheric CO2 being introduced to waters with naturally low levels of aragonite. Long term implications of acifdification can also be inferred from studying extreme natural habitats such as that detailed below.

This evidence, that ocean acidification is already impacting marine populations, is daunting and brings home the short time frame we have to alter our actions before more widespread effects are felt.
As of October 2012, atmospheric CO2 concentrations were measured as 391.03ppm (parts per million). According to the Intergovernmental Panel on Climate Change, this concentration will more than double to 800ppm by the year 2100 if we continue ‘business as normal’ i.e. burn fossil fuels at our current per capita rate.
To put this into perspective, the alarming 2050 computer model detailed above is based on carbon levels at a relatively modest 500ppm. If atmospheric CO2 concentrations were to reach 800ppm, many marine ecosystems would already have disappeared.
As with global warming, the only current solution is to reduce, and preferably reverse, greenhouse gas emissions. If CO2 in the atmosphere drops, there will be a net diffusion of CO2 out of the oceans and the ocean’s pH will be slowly restored.
Https://www.annualreviews.org/eprint/QwPqRGcRzQM5ffhPjAdT/full/10.1146/annurev.marine.010908.163834
Https://www.nature.com/ngeo/journal/vaop/ncurrent/full/ngeo1635.html#