Darren Naish is a fantastic advocate of everything to do with tetrapod zoology through his excellent blog. From fossils to modern day creatures, and the mysteries of cryptozoology (I recommend you look at the Loch Ness monster blog), Darren has an unquenchable thirst for knowledge of all things with four legs, or that have had four legs at some point in their evolutionary history. He has recently turned his attentions to giant salamanders and I hope you will enjoy his fascinating account of the cryptobranchids and the pictures that appear on his blog. The Chinese giant salamander is ranked 2 on the EDGE Amphibians list and is our highest priority amphibian for conservation action so this species is particularly close to our hearts…
Close up to Andrias, despite the smell and the teeth

Aww, look at that cute little face, those piggy little, opaque eyes, that wrinkled skin. I just know that you want a little refresher on giant salamanders, so – accompanied with new photos taken at the SMNK in Karlsruhe (by Markus Bühler; thanks) – here’s a substantially augmented chunk of text that originally appeared here back in January 2008…
Giant salamanders (or cryptobranchids) are grouped with hynobiids (Asiatic salamanders) in the clade Cryptobranchoidea (or Cryptobranchiformes). There are only three extant species: the North American Hellbender Cryptobranchus alleganiensis, the Chinese giant salamander Andrias davidianus and the Japanese giant salamander A. japonicus. All are aquatic salamanders of fast-flowing, well-oxygenated water (but this wasn’t the case for all fossil species, some of which apparently inhabited large ponds and lakes (Estes 1981, Tempfer 2004)). All possess dorsoventrally flattened bodies and flat heads with rounded snouts. Gills are absent in adults and their lungs apparently don’t function in respiration, so all gas exchange occurs across the extensively folded, wrinkled skin. Eyelids are absent (a sure sign of aquatic habits in a caudate). The following text is Andrias-heavy, though some of the generalisations apply to hellbenders too.

While the name currently in use for Asian giant salamanders (and their fossil relatives) is Andrias, the equally old Megalobatrachus has also been used a lot. Both were created by the same author (J. J. v. Tschudi), in the same year (1837), in the same published work, but Andrias was traditionally restricted to fossils and Megalobatrachus was traditionally restricted to the living species. Westphal (1958) argued that both should be synonymised, with Andrias taking priority. Some authors have regarded Andrias giant salamanders and hellbenders as part of the same genus (in part because they’ve regarded Andrias as paraphyletic with respect to Cryptobranchus), and have therefore used the name Cryptobranchus Leuckart, 1821 for the whole lot (Naylor 1981). More recent work has found both Andrias and Cryptobranchus to be monophyletic; fossils indicate that they had diverged by the Paleocene.
‘Mystery’ giant salamanders of California and Hong Kong
As any aficionado of the cryptozoological literature knows, there are some claimed occurrences of modern day giant salamanders that are very interesting, if real.
In 1939 or 1940, Stanford University’s George S. Myers was invited to examine a live Andrias kept as a pet by a commercial fisherman. Said fisherman claimed to have captured the animal in a catfish trap in the Sacramento River. Myers considered the possibility that the animal might belong to an unknown, native Californian species: he thought that the brownish (rather than grey) ground colour and presence of large, well defined, yellowish spots on the dorsum made it look different from Chinese and Japanese giant salamanders (Myers 1951). In fact, both features are present in Chinese and Japanese giant salamanders, something not noted by the cryptozoologists who have discussed this case. Myers also noted, however, that the animal might have originated as an escape: in 1936 or 1937, a San Francisco firm had supposedly imported a consignment of giant salamanders. In a brief report that smacks of ‘better late than never’, Chico State College’s Thomas L. Rodgers (1962) stated that he had examined the Sacramento River salamander two days after it had been captured. According to Rodgers, it belonged to an “odd-fish fancier” called Wong Hong and was named Benny. Rodgers regarded this as the end of the mystery, and ‘escaped pet’ is the generally accepted explanation today.
As Rodgers (1962) and others have explained, however, additional people claimed to see and even capture gigantic salamanders in the Californian Trinity Alps. An animal handler called Vern Harden said that he saw “a dozen” of these animals in Hubbard Lake in 1960, one of which he hooked and (allegedly) found to be 2.5 m long. During the 1920s, Frank L. Griffith reported seeing a group of giant salamanders in New River, the biggest of which was 2.75 m long (Rodgers 1962, Coleman 1997, Coleman & Huyghe 2003). Most recently, sightings from 2002 and 2005 were mentioned by cryptozoologist John Kirk.
As is so often the case with purported mystery animals, the possibility that northern California might be home to an undiscovered native amphibian approaching or exceeding a metre or two or three in length must rank as pretty low on the believability scale. Several qualified herpetologists have been interested enough to investigate the reports and, so far, have ended up empty handed. Rodgers suggested that, if the stories were true, they might be based on sightings of large Dicamptodon (Pacific giant salamanders), though individuals larger than just 30 cm would be exceptional [the above reconstruction of a Californian giant salamander – look at the scale! – is by Harry Trumbore and comes from Coleman & Huyghe (2003). Yeah, too many fingers]. After writing all this I discovered that Lord Geekington covered Californian giant salamanders in rather more depth back in March 2007.
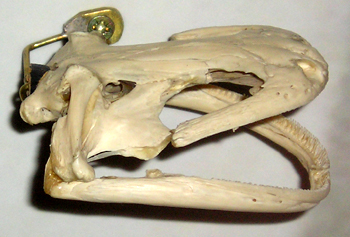
An entirely different ‘mystery giant salamander’ has been reported from Hong Kong where one was captured in the Botanical Gardens, Mid Levels, Hong Kong Island in 1922. This specimen was taken to London Zoo where it lived for many years; photos of it can be seen in zoo guide books published during the 1930s. In 1924, Edward G. Boulenger described it as representing the distinct species Megalobatrachus sligoi: it was supposedly distinct from the others on account of its smoother skin and flatter head (Boulenger 1924). It’s been stated that additional specimens were later captured, though the details on these are extremely hazy. It’s generally accepted today that Sligo’s salamander is synonymous with A. davidianus, though I’ve been unable to discover whether the individual(s) concerned were possible natives or the result of introduction (let me know if you have more information); the species is missing from field guides to the Hong Kong herpetofauna (e.g., Karsen et al. 1986) (thanks to Jon Downes, CFZ, for pers. comms.) [A. japonicus skeleton shown below].
Little known is that Chinese giant salamanders have also been found on Taiwan: these were almost definitely the result of introduction (people have moved Chinese giant salamanders around quite a bit: see below). Their presence in Vladivostok (in far eastern Russia) has also been rumoured.
Giant size, and the fossil ones
Giant salamanders are famous for, well, being giant, with record-holding specimens of the Chinese giant salamander reaching 1.8 m and 65 kg. However, some fossil species were bigger (read on). Some authors state that the Japanese species is bigger than the Chinese one, but fail to provide the measurements to back this up.
Having mentioned fossil Andrias salamanders, the most famous of them all is A. scheuchzeri from the Upper Miocene of Oeningen, Germany [shown here, by Haplochromis, from wikipedia]. The holotype skeleton – discovered in or prior to 1725 – was first assumed to be that of a man killed in the biblical flood.
It seems incredible today that any person (especially a learned one) could be dumb enough to think that a salamander skeleton should be best interpreted as that of a human, but I suppose I have the benefit of neither regarding biblical mythology as real, nor of being alive in the 1700s. Swiss scientist Johann Jakob Scheuchzer labelled the fossil Homo diluvii testis in his publications of 1726 and 1731. Cuvier realised in about 1822 that it was actually a salamander, Friedrich Holl gave it the proper binomial Salamandra scheuchzeri in 1831, and the new generic name Andrias was given to the species in 1837.
Numerous additional specimens of this salamander are known from Oligocene, Miocene and Pliocene sediments of Germany, Austria and the Czech Republic. Some were described as representing additional taxa, such as A. tschudii Meyer, 1859 and Zaissanurus belajaevaei Tschernov, 1959. A. scheuchzeri is said to be osteologically indistinguishable from living A. davidianus and Estes (1981) treated the latter as a synonym of the former. This opinion has never caught on: Darrel Frost notes in Amphibian Species of the World that it should be better known among neontologists. If it’s valid, the giant salamander populations of eastern China are sorry relicts of a formerly widespread species. Fossils suggest that these salamanders were also present on Japan in the Pleistocene (Estes 1981), in which case A. davidianus/A. scheuchzeri and A. japonicus were once sympatric.
Throughout this article I’ve (generally) been careful not to refer to Andrias salamanders as ‘Asian giant salamanders’ or even (in view of European A. scheuchzeri fossils) as ‘Old World giant salamanders’. This is because a species from Upper Miocene sediments of Nebraska, named in 1917 for a partial dentary, also seems to be a member of this genus. It’s A. matthewi.
[Zdeněk Burian’s reconstruction of A. scheuchzeri is shown here. As always, it looks great, but it’s not accurate: Andrias salamanders are strictly aquatic].
A large number of additional Miocene bones have been referred to A. matthewi, some because they closely resemble the corresponding bones of Old World Andrias species, others because they were found in association with dentary bones resembling the holotype (it’s proportionally slender and rather straight compared to the dentaries of other Andrias salamanders, suggesting that this species had a wedge-shaped, rather than rounded, snout). Like A. scheuchzeri, A. matthewi seems to have inhabited still water ponds and lakes, and not fast-flowing streams and such (Estes 1981). Naylor (1981) described two enormous vertebrae (hyperbole intentional, but we are talking about salamanders here) from Saskatchewan that he referred to A. matthewi. With estimated total centrum lengths of 30 and 40 mm, the larger of these bones indicates a total length for the animal of 2.3 m
Some sources refer to A. matthewi as Cryptobranchus matthewi. This isn’t necessarily because it was deemed closer to hellbenders than to Old World giant salamanders; rather – as mentioned above – it’s based on Naylor’s (1981) lumping of all Andrias species into Cryptobranchus. An Eocene salamander from Wyoming, Piceoerpeton willwoodensis Meszoely, 1967, might be a stem-cryptobranchid (as might other fossil salamanders, some from the Cretaceous), and fossil hellbender species are known too.
Den-masters, breeding burrows, polygyny and male clutch-care
Andrias salamanders grow continuously after maturity: I know this claim is made about many tetrapods, but it seems to have been actually demonstrated for both hellbenders and Japanese giant salamanders. It means that members of any breeding population can vary substantially in size; a consequence is that small males can be ‘sneakers’ that successfully mate with females while avoiding the aggressive attention of larger, territory-holding males (Kawamichi & Ueda 1998). The cloaca protrudes in breeding males, enabling them to be sexed by eye [A. japonicus skull shown in image below].
During the breeding season, Andrias salamanders species build long, horizontal mud burrows (as much as 1.5 m long) below the water line. The males that do this – dubbed ‘den-masters’ by Kawamichi & Ueda (1998) – are the longest and heaviest individuals in their habitats. They lay in wait in the burrows, emerging to attack other males that come into the territory. Some males quickly flee, but others stay and fight, and many individuals (as many as 17, perhaps more) may congregate outside the burrow and aim to get inside and mate with visiting females. I knocked up the terrible cartoon below to illustrate a few salient features of Andrias breeding behaviour.
Little known is that Andrias has particularly vicious teeth and can inflict massive wounds: during fights males frequently sever digits, limbs, and bits of tails of rivals, and massive fatal slices across the neck – sometimes resulting in decapitation – are apparently not uncommon. A male Japanese giant salamander 30 cm long lost in a fight to a much larger (c. twice as large) den-master; the latter then ate the former. Outside the breeding season, they use these teeth to capture and subdue frogs, fish, crustaceans, insects and (occasionally) small mammals.
Visiting females enter the burrows, and emerge at a reduced weight (indicating successful spawning: they produce strings of 400-600 eggs). They sometimes indulge in a bit of egg cannibalism while in the den-master’s burrow, evidently snacking on the efforts of other females. The den-master then stays at the nest until the eggs have hatched (about 40-50 days after spawning). Obviously, the bigger the male, the better he is at defending the clutches from predators. Juveniles stay in the larval stage for four or five years (reaching about 20 cm in total length), and take about ten years to reach adulthood.
Also little known of Andrias species is that they exude a foul smell which has been partially likened to “the rankest public urinal crossed with that of stale sweat” (Brazil 1997, p. 64).
Variation in Andrias: low in Japan, weird in China
A low level of genetic variation has been reported for the Japanese giant salamander (Matsui & Hayashi 1992): this might be due to their sedentary behaviour and a mating system that involves nest-guarding and polygyny (conceivably, this strategy could mean that a few males might be responsible for re-stocking a given region after environmental disturbance). A population bottleneck resulting from end-Pleistocene decline might also be partly responsible (Matsui et al. 2008). Chinese giant salamanders exhibit more variation (with a population in Huangshan being particularly distinct). What’s interesting is that the observed variation among Chinese giant salamanders doesn’t match what you might predict given the drainage patterns of the relevant rivers. Accordingly, the distribution of populations might be the result of human translocations (Murphy et al. 2000). This means that any original phylogeographical pattern has all but been destroyed. I’d rather have interspecific hybrid giant salamanders than no giant salamanders at all, but these efforts have not been mindful of the original local genetic patterns [below 1887 illustration of a Japanese giant salamander from wikipedia].
And, the bad news…
The bad news is that these amazing animals – particularly the Chinese species – are critically endangered thanks to habitat degradation and loss, and also to human hunting. Yes, people EAT these animals, with their flesh selling for as much as US$100 per kg. Literally 1000s of kg of giant salamander flesh was harvested from Chinese provinces in decades past – as much as 15,000 kg per year from Hunan, for example. Unsurprisingly, there have been attempts to farm the animals, but little seems to be known about how successful (or otherwise) these ventures were.
Mass die-offs have been reported due to pollution (in 1986, 46 Japanese giant salamanders died after illegal dumping of oil). The declining hellbenders of the Ozarks are known to be infected by both the amphibian-killing chytrid fungus Batrachochytrium dendrobatidis (Bd) and the flesh-eating bacterium Citrobacter freundii, though as yet there’s no indication (to my knowledge) that Asian giant salamanders are affected by either organism. Also worth keeping in mind is that giant salamanders are sensitive to water-warming and other forms of disturbance. This was demonstrated by the case in October 2010 where 15 captive animals – kept at an exhibition in Shanghai – died “as a result of the hot weather and noisy environment”.
Mark-recapture studies of Japanese giant salamanders have shown that, even in areas with apparently healthy populations of large adults, successful recruitment of larvae to the adult population is very low, mostly because stream modification is destroying both spawning regions and larval habitats (Okada et al. 2008). There is loads more information on the conservation status and biology of the Chinese giant salamander here on the EDGE site (EDGE = Evolutionarily Distinct and Globally Endangered).
Amazingly, this article started life as ‘here are some photos of a giant salamander’. Oh well… I did always plan to cover these salamanders at some point, and better late than never. For more on salamanders at Tet Zoo, see the ‘group overview’ articles here…
- Giants and sirens: caudates part I
- The wonder that is the internally fertilizing salamander clade: caudates part II
And for more in-depth looks at particular clades and species, see…
- Spiky-frilled, lek-breeding amphibious salamanders… or ‘newts’
- Coprophagy and the giraffe-neck program: more on plethodontids
- Axolotls on the EDGE!
- When salamanders invaded the Dinaric Karst: convergence, history, and reinvention of the troglobitic olm
- The USA is still yielding lots of new extant tetrapod species
- Amphiumas: gigantism, extended parental care and freaky morphology in a group of eel-like salamanders
And for more on the Global Amphibian Crisis, see…
- Get ready for 2008: Year Of The Frog
- The EDGE amphibian project launches today
- California’s declining frogs
- It is still Year of the Frog
- A world without Baw Baw frogs?
- The Global Amphibian Crisis, 2009
Refs – –
Brazil, M. 1997. Mission massive. BBC Wildlife 15 (4), 62-67.
Boulenger, E. G. 1924. On a new giant salamander, living in the Society’s Gardens. Proceedings of the Zoological Society of London 1924, 173-174.
Coleman, L. 1997. Promises of giants. Fortean Times 103, 43.
– . & Huyghe, P. 2003. The Field Guide to Lake Monsters, Sea Serpents, and Other Mystery Denizens of the Deep. Tarcher/Penguin, New York.
Estes, R. 1981. Handbuch der Paläoherpetologie. Teil 2. Gymnophiona, Caudata. Gustav Fischer Verlag, Stuttgart.
Karsen, S. J., Lau, M. W.-n. & Bogadek, A. 1986. Hong Kong Amphibians and Reptiles. Urban Council, Hong Kong.
Kawamichi, T., & Ueda, H. (1998). Spawning at Nests of Extra-Large Males in the Giant Salamander Andrias japonicus Journal of Herpetology, 32 (1) DOI: 10.2307/1565495
Matsui, M. & Hayashi, T. 1992. Genetic uniformity in the Japanese giant salamander, Andrias japonicus. Copeia 1992, 232-235.
– ., Tominaga, A., Liu, W.-z. & Tanaka-Ueno, T. 2008. Reduced genetic variation in the Japanese giant salamander, Andrias japonicus (Amphibia: Caudata). Molecular Phylogenetics and Evolution 49, 318-326.
Murphy, R. W., Fu, J., Upton, D. E., de Lemas, T. & Zhao, E.-M. 2000. Genetic variability among endangered Chinese giant salamanders, Andrias davidianus. Molecular Ecology 9, 1539-1547.
Myers, G. S. 1951. Asiatic giant salamander caught in the Sacramento River, and an exotic skink near San Francisco. Copeia 1951, 179-180.
Naylor, B. G. 1981. Cryptobranchid salamanders from the Paleocene and Miocene of Saskatchewan. Copeia 1981, 76-86.
Okada, S., Utsunomiya, T., Okada, T., Felix, Z. I. & Ito, F. 2008. Characteristics of Japanese giant salamander (Andrias japonicus) populations in two small tributary streams in Hiroshima Prefecture, western Honshu, Japan. Herpetological Conservation and Biology 3, 192-202.
Rodgers, T. L. 1962. Report of giant salamanders in California. Copeia 1962, 646-647.
Tempfer, P. M. 2004. Andrias scheuchzeri (Caudata: Crytobranchidae) aus der obermiozänen (MN7/8) Fundstelle Mataschen/steiermark. Joannea Geol. Paläont. 5. 257-268.
Westphal, F. 1958. Die Tertiären und Rezenten Eurasiatischen Risensalamander (genus Andrias, Urodela, Amphibia). Palaeontographica 110, 20-92.